Thematic Eq E.Rubinstein - C.Boucheix - S.Charrin
Membrane compartmentalization by tetraspanins and cell differentiation
Scientific Team Leader: : Dr. Eric Rubinstein (DR Inserm)
Thematic
Tetraspanins play a crucial role in many physiological functions such as sperm-egg fusion, muscle regeneration, the visual and renal functions , as well as immunity. They also play an important role in major pathologies including cancer, malaria, hepatitis C and AIDS. They play a role in cell compartmentalization , through the regulation of the trafficking of various proteins, and also in membrane compartmentalization. The goal of the “tetraspanin” team is to determine how tetraspanins function at the molecular level, and to unravel the physiopathological importance of some of them.
Cells communicate with their environment through membrane receptors which recognize molecules and particles in the external milieu and transmit an intracellular signal that can lead to gene activation or alternatively to activation of other membrane receptors and cytoplasmic targets. Whereas the membrane has long been considered a fluid mosaic, most recent evidence show that it is not homogeneous, and that several factors oppose to the free diffusion of molecule in the membrane. One of these factors is the interaction with membrane structures of particular composition such as microdomains. Membrane compartmentalization inside these microdomains contributes to ligand binding and/or signalling.
We have been interested in membrane compartmentalization through the study of a family of membrane proteins called tetraspanins. Tetraspanins are four transmembrane domains proteins with discrete structural features. There are 33 tetraspanins in mammals, and so far all cell types studied express several of these molecules, often at a high level. Tetraspanins have been implicated in various biological processes such as cell adhesion, migration, cell fusion, co-stimulation, signal transduction, and differentiation. The array of observed effects suggest that tetraspanins play a role in basal functions of the cell.
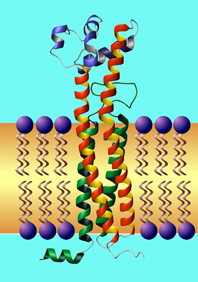
Fig.1: the structure of a typical tetraspanin, according to the modelling of Michel Seigneuret.
The importance of these molecules in various physiological and pathological conditions has been unravelled during the recent years. There are now several genetic diseases caused by mutations in particular tetraspanins. Also several tetraspanins are used for the infection by several pathogens. In particular, HIV recruits several tetraspanins at the site of budding. The tetraspanin CD81 drew particular attention because it plays an essential role in the infection by two important human pathogens. It is on the one hand an obligatory receptor for the hepatitis C virus, a major cause of hepatocellular carcinoma. Additionally, we have shown in collaboration with Olivier Silvie and Dominique Mazier (Inserm U1135) that in plays a major role in the entry of the malaria parasite into hepatocytes (Silvie et al., Nat. Med., 2003, 9: 93).
The idea that tetraspanins play a role in membrane compartmentalization, that could be defined as non-randomness (of localization, of interactions) in the membrane, comes from the demonstration by several groups, including ours, that tetraspanins organize a network of interactions that we referred to as the tetraspanin web (Rubinstein et al., Eur. J. Immunol., 1996, 26: 2657) (Rubinstein et al., Eur. J. Immunol., 1996, 26: 2657), and is also called as tetraspanin-enriched microdomains. We have shown that these structures are highly organized, with a particular tetraspanin targeting specific non-tetraspanin partner molecules (to which they associate directly) to these domains. We have demonstrated that they are built with the help of lipids, cholesterol participating to the maintenance of these structures, as well as lipid-modifications (palmitoylation) of tetraspanins (Charrin et al., FEBS Lett., 2002, 516: 139; Charrin et al., Eur. J. Immunol., 2003, 33: 2479). We have also published the first proteomic characterization of these structures: They contain 4 major groups of proteins, including integrins, proteins with Ig domains, ectoenzymes and heterotrimeric G proteins (Andre et al., Proteomics., 2006, 6: 1437; Le Naour et al., Mol. Cell Proteomics., 2006, 5: 845).

Fig. 2 Les tétraspanines constituent un puzzle moléculaire à la surface cellulaire:
Biochemical studies have demonstrated specific interactions of certain tetraspanins (here in green) with a few numbers of associated molecules (their molecular partners). For example, CD9 and CD81 have two common partners designed as CD9P-1 or EWI-2. Discrete microdomains arise as tetraspanins associate with each others. Thus each tetraspanin is likely to induce the association of its molecular partners with the microdomains.
Our current projects concern three axis of research based on the studies we have published in the recent years.
Axe 1
The role of the tetraspanins in intercellular fusion process.
We have in the past demonstrated, through the creation of knock-out mice, that the tetraspanin CD9 present on the oocyte surface plays an essential role in sperm-egg fusion, in cooperation with CD81 (Le Naour et al., Science, 2000, 287: 319; Rubinstein et al., Dev. Biol., 2006, 290: 351). We are still analyzing the role of CD9 in this process, in collaboration with Christine Gourier and Eric Perez, at ENS Paris, et the team of Jean-Philippe Wolf at Cochin institute.
We have also recently shown that these two tetraspanins, CD9 and CD81, are negative regulators of the fusion of muscle cells (Charrin et al., 2013, Nat Commun.;4:1674). Their presence prevents the fusion of multinucleated myotubes in vitro, and of regenerating myofibers in vivo. This negative regulation of fusion plays a role in the ad integrum restoration of muscle architecture in the course of regeneration. We have also demonstrated that CD9P-1, a direct partner of both CD9 and CD81 contributed to this negative regulation of fusion. We are studying the underlying mechanisms.
In conclusion, our study unveils a mechanism preventing inappropriate fusion of myotubes that plays an important role in the restitution of normal muscle architecture during muscle regeneration.
Axet 2
Tspan8 in colon cancer.
The tetraspanin Tspan8 has a restricted pattern of expression, and is particularly expressed on digestive epitheliums. We have shown on the one hand that a decrease expression of Tspan8 is a good prognosis parameter in colon cancer. On the other hand, Tspan8 coordinates cell motility by integrating various signals issued from the membrane (integrins, E-cadherin, EGFR) (Greco et al., 2010, Cancer Res 70 :7674). In addition, we have shown that the anti-Tspan8 mAb TS29.2 produced in the laboratory significantly inhibited the growth of colon cancer cell lines grafted in the mouse (Ailane et al., 2014, Front Physiol., 5:364).
Ainsi ce travail démontre que la tétraspanine Tspan8 joue un rôle important dans la progression du cancer du côlon et suggère qu’il pourrait constituer une bonne cible thérapeutique dans cette pathologie.
Axe 3
Regulation of the metalloprotease ADAM10 by tetraspanins and its implication for Notch signalling.
We have shown that a subgroup of 6 tetraspanins (Tspan5, 17, 14, 33, 5, 10) that have 8 cysteines in the large extracellular domain (and therefore are referred to as TspanC8) were all direct partners of ADAM10 and regulated its exit from the endoplasmic reticulum (Dornier, et al., 2012, J Cell Biol,199:481).
ADAM10 plays a key role in Notch signaling. Indeed, Notch can be described as a membrane-tethered transcriptional co-activator . The binding of Notch to its membrane-tethered ligands results in a double cleavage by ADAM10 and the γ-secretase complex that allow the translocation of Notch intracellular domains into the nucleus. We have shown that Tspan5 and Tspan15 are positive regulators of Notch signaling, probably by regulating ADAM10 ability to cleave Notch.
En collaboration avec l'équipe de François Schweisguth (Institut Pasteur), nous avons montré que les TspanC8 de drosophile s'associent également à ADAM10 (aussi appelée Kuz chez la drosophile) dont elles modulent la distribution cellulaire, régulent l'expression de cibles de Notch, et jouent un rôle important dans plusieurs aspects du développement dépendant de Notch.
Most mammalian tetraspanins have no orthologs in invertebrates, suggesting an independent evolution of these molecules in the different phylums. In contrast, TspanC8 have clear orthologs in C. elegans and D. melanogaster (up to 40 % identity). We have shown, in collaboration with the group of François Schweisguth (Institut Pasteur), that the three Drosophila TspanC8 modulate Notch signalling in vivo, acting specifically on the receptor, and that this was associated with a modification of Kuz localization.
In conclusion, this study has identified a set of evolutionary conserved tetraspanins that control ADAM10 surface expression level thereby regulating Notch signaling.
Contacts :
Eric Rubinstein : This email address is being protected from spambots. You need JavaScript enabled to view it.
Stéphanie Charrin : This email address is being protected from spambots. You need JavaScript enabled to view it.
Claude Boucheix : This email address is being protected from spambots. You need JavaScript enabled to view it.
Further reading :
Boucheix C, Rubinstein E. Tetraspanins. Cell Mol Life Sci. 2001 Aug;58(9):1189-205.
Pubications
2015
Desterke C, Martinaud C, Guerton B, Pieri L, Bogani C, Clay D, Torossian F, Lataillade JJ, Hasselbalch HC, Gisslinger H, Demory JL, Dupriez B, Boucheix C, Rubinstein E, Amsellem S, Vannucchi AM, Le Bousse-Kerdilès MC. The tetraspanin CD9 participates in dysmegakaryopoiesis and in stroma interactions in primary myelofibrosis. Haematologica. 2015 Apr 3. pii: haematol.2014.118497. [Epub ahead of print]
Martínez Del Hoyo G, Ramírez-Huesca M, Levy S, Boucheix C, Rubinstein E, Minguito de la Escalera M, González-Cintado L, Ardavín C, Veiga E, Yáñez-Mó M, Sánchez-Madrid F. CD81 Controls Immunity to Listeria Infection through Rac-Dependent Inhibition of Proinflammatory Mediator Release and Activation of Cytotoxic T Cells.J Immunol. 2015 May 13. pii: 1402957. [Epub ahead of print]
2014
Chalbi M, Barraud-Lange V, Ravaux B, Howan K, Rodriguez N, Soule P, Ndzoudi A, Boucheix C, Rubinstein E, Wolf JP, Ziyyat A, Perez E, Pincet F, Gourier C. Binding of sperm protein Izumo1 and its egg receptor Juno drives Cd9 accumulation in the intercellular contact area prior to fusion during mammalian fertilization. Development. 2014 Oct;141(19):3732-9
Ailane N, Greco C, Zhu Y, Sala-Valdés M, Billard M, Casal I, Bawa O, Opolon P, Rubinstein E, Boucheix C. Effect of an anti-human Co-029/tspan8 mouse monoclonal antibody on tumor growth in a nude mouse model. Front Physiol. 2014 Sep 19;5:364.
Charrin S, Jouannet S, Boucheix C, Rubinstein E. Tetraspanins at a glance. J Cell Sci. 2014 Sep 1;127(Pt 17):3641-8.
Dahmane S, Rubinstein E, Milhiet PE. Viruses and tetraspanins: lessons from single molecule approaches. Viruses. 2014 May 5;6(5):1992-2011.
2013
Risco-Castillo V, Son O, Franetich JF, Rubinstein E, Mazier D, Silvie O. Phase hépatique du paludisme : voies d’entrée des sporozoïtes de Plasmodium. Biol Aujourdhui. 2013;207(4):219-29. doi: 10.1051/jbio/2013021.
Charrin S, Latil M, Soave S, Polesskaya A, Chrétien F, Boucheix C, Rubinstein E. Normal muscle regeneration requires tight control of muscle cell fusion by tetraspanins CD9 and CD81. Nat Commun. 2013 Apr 9;4:1674. doi: 10.1038/ncomms2675.
Potel J, Rassam P, Montpellier C, Kaestner L, Werkmeister E, Tews BA, Couturier C, Popescu CI, Baumert TF, Rubinstein E, Dubuisson J, Milhiet PE, Cocquerel L. EWI-2wint promotes CD81 clustering that abrogates Hepatitis C Virus entry. Cell Microbiol. 2013 Jul;15(7):1234-52. doi: 10.1111/cmi.12112. Epub 2013 Feb 16.
Rubinstein E, Charrin S, Tomlinson MG. Organisation of the Tetraspanin Web. In Tetraspanins, F. Berditchevski, and E. Rubinstein, eds. (Springer Netherlands), Series: Proteins and Cell Regulation, 2013, Vol. 9 pp. 47–90.
Copeland BT, Bowman MJ, Boucheix C, Ashman LK. Knockout of the tetraspanin Cd9 in the TRAMP model of de novo prostate cancer increases spontaneous metastases in an organ-specific manner. Int J Cancer. 2013 Oct 15;133(8):1803-12. doi: 10.1002/ijc.28204. Epub 2013 May 15
Obeid M, Franetich JF, Lorthiois A, Gego A, Grüner AC, Tefit M, Boucheix C, Snounou G, Mazier D. Skin-draining lymph node priming is sufficient to induce sterile immunity against pre-erythrocytic malaria. EMBO Mol Med. 2013 Feb;5(2):250-63. doi: 10.1002/emmm.201201677. Epub 2012 Dec 19.
2012
Dornier E, Coumailleau F, Ottavi JF, Moretti J, Boucheix C, Mauduit P, Schweisguth F and Rubinstein E. TspanC8 tetraspanins regulate the trafficking of ADAM10/Kuzbanian and Notch receptor activation in flies and mammals. J Cell Biol. 2012, 199:481-496.
Sala-Valdés M, Ailane N, Greco C, Rubinstein E, Boucheix C. Targeting tetraspanins in cancer. Expert Opin Ther Targets. 2012 Oct;16(10):985-97.
2011
van Niel G, Charrin S, Simoes S, Romao M, Rochin L, Saftig P, Marks MS, Rubinstein E, Raposo G. The Tetraspanin CD63 Regulates ESCRT-Independent and -Dependent Endosomal Sorting during Melanogenesis. Dev Cell. 2011, 21: 708-721.
Rubinstein E. The complexity of tetraspanins. Biochem Soc Trans. 2011 Apr 1;39(2):501-5.
He B, Zhang YH, Richardson MM, Zhang JS, Rubinstein E, Zhang XA. Differential functions of phospholipid binding and palmitoylation of tumor suppressor EWI-2. Biochem J. 2011 Aug 1;437(3):399-411.
Montpellier C, Tews BA, Poitrimole J, Rocha-Perugini V, D'Arienzo V, Potel J, Zhang XA, Rubinstein E, Dubuisson J, Cocquerel L. Interacting regions of CD81 and two of its partners, EWI-2 and EWI-2wint and their effect on Hepatitis C Virus infection. J Biol Chem. 2011 Apr 22;286(16):13954-65.
Bollée G, Flamant M, Schordan S, Fligny C, Rumpel E, Milon M, Schordan E, Sabaa N, Vandermeersch S, Galaup A, Rodenas A, Casal I, Sunnarborg SW, Salant DJ, Kopp JB, Threadgill DW, Quaggin SE, Dussaule JC, Germain S, Mesnard L, Endlich K, Boucheix C, Belenfant X, Callard P, Endlich N, Tharaux PL. Epidermal growth factor receptor promotes glomerular injury and renal failure in rapidly progressive crescentic glomerulonephritis. Nat Med. 2011 Sep 25;17(10):1242-50. doi: 10.1038/nm.2491. Erratum in: Nat Med. 2011 Oct;17(10):2
2010
Greco C, Bralet MP, Ailane N, Dubart-Kupperschmitt A, Rubinstein E, Le Naour F and Boucheix C. E-cadherin/p120-catenin and tetraspanin Co-029 cooperate for cell motility control in human colon carcinoma. Cancer Res. 2010 Oct 1;70(19):7674-83.
Cailleteau L, Estrach S, Thyss R, Boyer L, Doye A, Domange B, Johnsson N, Rubinstein E, Boucheix C, Ebrahimian T, Silvestre JS, Lemichez E, Meneguzzi G, Mettouchi A. alpha2beta1 integrin controls association of Rac with the membrane and triggers quiescence of endothelial cells. J Cell Sci. 2010 Jul 15;123(Pt 14):2491-501.
Tham TN, Gouin E, Rubinstein E, Boucheix C, Cossart P, Pizarro-Cerda J. Tetraspanin CD81 is required for Listeria monocytogenes invasion. Infect Immun. 2010 Jan;78(1):204-9.
2009
Wakabayashi T, Craessaerts K, Bammens L, Bentahir M, Borgions F, Herdewijn P, Staes A, Timmerman E, Vandekerckhove J, Rubinstein E, Boucheix C, Gevaert K, De Strooper B. Analysis of the gamma-secretase interactome and validation of its association with tetraspanin-enriched microdomains. Nat Cell Biol. 2009 Nov;11(11):1340-6.
Charrin S, Yalaoui S, Bartosch B, Cocquerel L, Franetich JF, Boucheix C, Mazier D, Rubinstein E*, Silvie O*. The Ig domain protein CD9P-1 down-regulates CD81 ability to support Plasmodium yoelii infection. J Biol Chem. 2009 Nov 13;284(46):31572-8. (* equal contribution, corresponding authors)
André M, Chambrion C, Charrin S, Soave S, Chaker J, Boucheix C, Rubinstein E and Le Naour F. In situ chemical cross-linking on living cells reveals CD9P-1 cis-oligomer at cell surface. J Proteomics. 2009 Nov 2;73(1):93-102.
Charrin S, Le Naour F., Silvie O, Milhiet PE, Boucheix C and Rubinstein E. Lateral organization of membrane proteins: tetraspanins spin their web. Biochem J. 2009 May 13;420(2):133-54.
Rocha-Perugini V, Lavie M, Delgrange D, Canton J, Pillez A, Potel J, Lecoeur C, Rubinstein E, Dubuisson J, Wychowski C, Cocquerel L.. The association of CD81 with tetraspanin-enriched microdomains is not essential for Hepatitis C Virus entry. BMC Microbiology 2009, 9:111
Nakamoto T, Murayama Y, Oritani K, Boucheix C, Rubinstein E, Nishida M, Katsube F, Watabe K, Kiso S, Tsutsui S, Tamura S, Shinomura Y, Hayashi N. A novel therapeutic strategy with anti-CD9 antibody in gastric cancers. J Gastroenterol. 2009;44(9):889-96
Klibi J, Niki T, Riedel A, Pioche-Durieu C, Souquere S, Rubinstein E, Moulec SL, Guigay J, Hirashima M, Guemira F, Adhikary D, Mautner J, Busson P. Blood diffusion and Th1-suppressive effects of galectin-9-containing exosomes released by Epstein-Barr virus-infected nasopharyngeal carcinoma cells. Blood. 2009 Feb 26;113(9):1957-66. Epub 2008 Nov 12.
2008
Blood diffusion and Th1-suppressive effects of galectin-9-containing exosomes released by Epstein-Barr virus-infected nasopharyngeal carcinoma cells. Klibi J, Niki T, Riedel A, Pioche-Durieu C, Souquere S, Rubinstein E, Le Moulec S, Guigay J, Hirashima M, Guemira F, Adhikary D, Mautner J, Busson P. Blood. 2008 Nov 12. [Epub ahead of print]
Arduise C, Abache T, Lei Li, Billard M, Chabanon A, Ludwig A, Mauduit P, Boucheix C, Rubinstein E*, Le Naour F*. Tetraspanins regulate ADAM10-mediated cleavage of TNFα and EGF. J. Immunol. 2008, 181: 7002-7013. (* Contribution identique)
Espenel C , Margeat E, Dosset P, Arduise C, Le Grimellec C, Royer CA, Boucheix C, Rubinstein E, Milhiet PE. Single molecule analysis of CD9 dynamics and partitioning reveals multiple modes of interaction in the tetraspanin web. J Cell Biol. 2008, 182(4):765-76.
Yamada M, Tamura Y, Sanzen N, Sato-Nishiuchi R, Hasegawa H, Ashman LK, Rubinstein E, Yáñez-Mó M, Sánchez-Madrid F, Sekiguchi K. Probing the interaction of tetraspanin CD151 with integrin alpha3beta1 using a panel of monoclonal antibodies with distinct reactivities toward the CD151-integrin alpha3beta1 complex. Biochem J. 2008, 415(3):417-27
Reman O, Pigneux A, Huguet F, Vey N, Delannoy A, Fegueux N, de Botton S, Stamatoullas A, Tournilhac O, Buzyn A, Charrin C, Boucheix C, Gabert J, Lhéritier V, Vernant JP, Fière D, Dombret H, Thomas X. Central nervous system involvement in adult acute lymphoblastic leukemia at diagnosis and/or at first relapse: Results from the GET-LALA group. Leuk Res. 2008 Nov;32(11):1741-50.
Tamgüney G, Giles K, Glidden DV, Lessard P, Wille H, Tremblay P, Groth DF, Yehiely F, Korth C, Moore RC, Tatzelt J, Rubinstein E, Boucheix C, Yang X, Stanley P, Lisanti MP, Dwek RA, Rudd PM, Moskovitz J, Epstein CJ, Cruz TD, Kuziel WA, Maeda N, Sap J, Ashe KH, Carlson GA, Tesseur I, Wyss-Coray T, Mucke L, Weisgraber KH, Mahley RW, Cohen FE, Prusiner SB. Genes contributing to prion pathogenesis. J Gen Virol. 2008 Jul;89(Pt 7):1777-88.
Yalaoui S., Zougbédé S., Charrin S., Silvie O., Arduise C., Farhati K., Boucheix C., Mazier D., *Rubinstein E. *Froissard P. Hepatocyte permissiveness to plasmodium infection is conveyed by a short and structurally conserved region of the CD81 large extracellular domain. PLOS Pathogen, 2008,4(2)1-12. (*equal contribution)
Rocha-Perugini V, Montpellier C, Delgrange D, Wychowski C, Helle F, Pillez A, Drobecq H, Le Naour F, Charrin S, Levy S, Rubinstein E, Dubuisson J, Cocquerel L. The CD81 partner EWI-2wint inhibits hepatitis C virus entry. PLoS ONE. 2008 Apr 2;3(4):e1866.
Reman O, Pigneux A, Huguet F, Vey N, Delannoy A, Fegueux N, de Botton S, Stamatoullas A, Tournilhac O, Buzyn A, Charrin C, Boucheix C, Gabert J, Lhéritier V, Vernant JP, Fière D, Dombret H, Thomas X. Central nervous system involvement in adult acute lymphoblastic leukemia at diagnosis and/or at first relapse: Results from the GET-LALA group. Leuk Res. 2008 May 26. PMID: 18508120.
2007
Silvie O, Franetich JF, Boucheix C, Rubinstein E, Mazier D. Alternative invasion pathways for plasmodium berghei sporozoites. Int J Parasitol. 2007;37:173-182.
Tavernier E, Boiron J-M, Huguet F, Bradstock K, Vey N, Kovacsovics T, Delannoy A, Fegueux N, Fenaux P, Stamatoullas A, Tournilhac O, Buzyn A, Reman O, Charrin C, Boucheix C, Gabert J, Lheritier V, Vernant J-P, Dombret H and Thomas X, for the GET-LALA Group, the Swiss Group for Clinical Cancer Research SAKK and the AustralasianLeukaemia and Lymphoma Group. Outcome of treatment after first relapse in adults with acute lymphoblastic leukaemia initially treated by the LALA-94 trial. Leukemia 2007, 21:1907-1914.
Abache T, Le Naour F, Planchon S, Harper F, Boucheix C, Rubinstein E. The transferrin receptor and the tetraspanin web molecules CD9, CD81, and CD9P-1 are differentially sorted into exosomes after TPA treatment of K562 cells. J Cell Biochem. 2007 Oct 15;102(3):650-664.
André M., Morelle W., Planchon S., Milhiet P.E., Rubinstein E., Mollicone R, Chamot-Rooke J. and Le Naour F. Glycosylation status of the membrane protein CD9P-1. Proteomics 2007, 7 :3880–3895
Le Naour F. Identification of tumor antigens by using proteomics. Methods in Molecular Biology, vol., 360, Target Discovery and Validation: Reviews and Protocols. Emerging Strategies for Targets and Biomarker Discovery, Humana Press Inc., Totowa, NJ. 2007, vol I: 327-334.
2006
Silvie O, Greco C, Franetich J-F, Dubart-Kupperschmitt A, Hannoun L, van Gemert G-J, Sauerwein R W, Levy S, Boucheix C, Rubinstein E and Mazier D. Expression of human CD81 differently affects host cell susceptibility to malaria sporozoites depending on the Plasmodium species. Cellular Microbiology 2006, 8 :1134-1146.
Stoeck A, Keller S, Riedle S, Sanderson MP, Runz S, Le Naour F, Gutwein P, Ludwig A, Rubinstein E, Altevogt P. A role for exosomes in the constitutive and stimulus-induced ectodomain cleavage of L1 and CD44. Biochem J. 2006, 393:609-618.
Ziyyat A*, Rubinstein E*, Monier-Gavelle F., Barraud V., Kulski O., Prenant M., Boucheix C., Bomsel M., and Wolf J.-P.CD9 controls the formation of clusters containing tetraspanins and the integrin α6β1 involved in human and mouse gamete fusion. J. Cell Science, 2006, 119:416-424. (*equal contribution)
Dhedin N, Dombret H, Thomas X, Lheritier V, Boiron JM, Rigal-Huguet F, Vey N, Kuentz M, Reman O, Witz F, Delannoy A, Kovacsovics T, Bradstock K, Charrin C, Boucheix C, Gabert J, Blaise D, Fiere D, Vernant JP. Autologous stem cell transplantation in adults with acute lymphoblastic leukemia in first complete remission: analysis of the LALA-85, -87 and -94 trials. Leukemia. 2006, 20:336-344.
Chometon G., Zhi-Gang Zhang, Rubinstein E., Boucheix C, Mauch C., Aumailley M. Dissociation of the complex between CD151 and laminin-binding integrins permits migration of epithelial cells Exp Cell Res. 2006, 312:983-995.
Rubinstein E, Ziyyat A, Prenant M, Wrobel E, Wolf JP, Levy S, Le Naour F, Boucheix C. Reduced fertility of female mice lacking CD81. Dev Biol. 2006, 290:351-358.
Le Naour F, André M, Greco C, Billard M, Sordat B, Emile JF, Lanza F, Boucheix C, Rubinstein E. Profiling of the tetraspanin web of human colon cancer cells. Mol Cell Proteomics. 2006, 5:845-857.
André M.,. Le Caer J.-P., Greco C., Planchon S., El Nemer W., Boucheix C, Rubinstein E., Chamot-Rooke J. and Le Naour F. Proteomic analysis of the tetraspanin web using LC-ESI-MS/MS and MALDI-FTICR-MS Proteomics, 2006, 6:1437-1449.
Silvie O, Charrin S, , Billard M, Franetich JF, Clark K, van Gemert GJ, Sauerwein RW, Dautry F, Boucheix C, Mazier D, Rubinstein E. Cholesterol contributes to the organization of tetraspanin-enriched microdomains and to CD81-dependent infection by malaria sporozoites. J. Cell Science, 2006, 119 :1992-2002.
Gego A , Silvie O, Franetich JF, Farhati K, Hannoun, Luty AF, Sauerwein RW, Boucheix C, Rubinstein E, Mazier D. A new approach for high-throughput screening of drug activity on Plasmodium liver stages. Antimicrobial Agents and Chemotherapy, 2006, 50:1586-1589.
Sala-Valdes M, Ursa A, Charrin S, Rubinstein E, Hemler ME, Sanchez-Madrid F, Yanez-Mo M. EWI-2 and EWI-F link the Tetraspanin Web to the actin cytoskeleton through their direct association with Ezrin-Radixin-Moesin proteins. J Biol Chem. 2006, 281:19665-75.
Yang XH, Kovalenko OV, Kolesnikova TV, Andzelm MM, Rubinstein E, Strominger JL, Hemler ME. Contrasting effects of EWI proteins, integrins, and protein palmitoylation on cell surface CD9 organization. J Biol Chem. 2006, 281:12976-85.
Picard C, Hayette S, Bilhou-Nabera C, Cayuela JM, Delabesse E, Frenoy N, Preudhomme C, Dupont M, Bastard C, Bories D, Vaerman JL, Davi F, Dastugue N, Raynaud S, Lafage M, Deschaseaux F, Fest T, Gaub MP, Lheritier V, Thomas X, Charrin C, Boucheix C, Dombret H, Macintyre E, Fiere D, Gabert J. Prospective multicentric molecular study for poor prognosis fusion transcripts at diagnosis in adult B-lineage ALL patients: the LALA 94 experience. Leukemia. 2006 ; 20 :2178-81
Vey N, Thomas X, Picard C, Kovascovicz T, Charin C, Cayuela JM, Dombret H, Dastugue N, Huguet F, Bastard C, Stamatoulas A, Giollant M, Tournilhac O, Macintyre E, Buzyn A, Bories D, Kuentz M, Dreyfus F, Delannoy A, Raynaud S, Gratecos N, Bordessoule D, de Botton S, Preudhomme C, Reman O, Troussard X, Pigneux A, Bilhou C, Vernant JP, Boucheix C, Gabert J. Allogeneic stem cell transplantation improves the outcome of adults with t(1;19)/E2A-PBX1 and t(4;11)/MLL-AF4 positive B-cell acute lymphoblastic leukemia: results of the prospective multicenter LALA-94 study. Leukemia. 2006; 20:2155-61
